Bayer Adempas (riociguat) Phase IV Study Is Successful: Strong Efficacy In Patients With Insufficient PDE5i Response! - Industry news - News - Hefei Home Sunshine Pharmaceutical Technology Co., Ltd

Riociguat. Comprimido. Cada comprimido contiene: Riociguat 2.5 mg Envase con 42 comprimidos. - Heka – Insumos Medicos
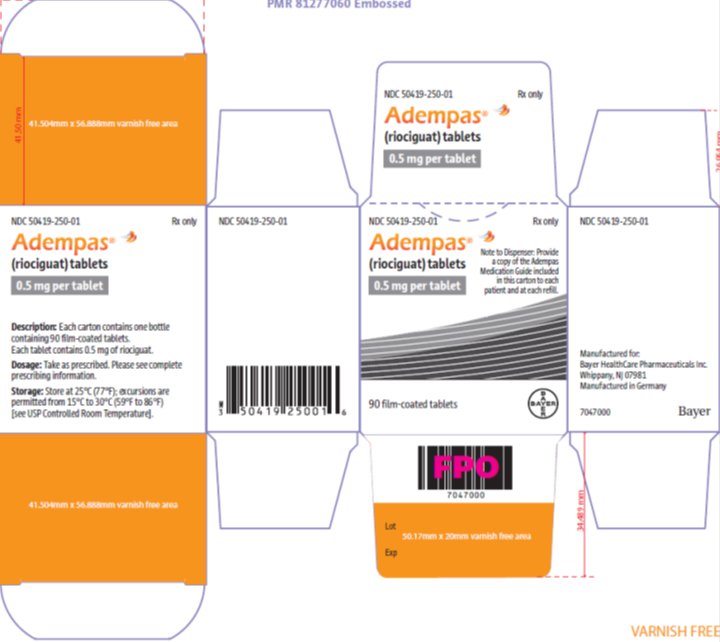
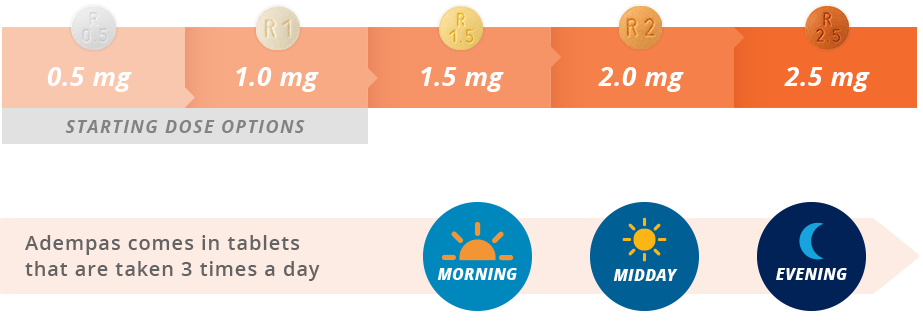
These highlights do not include all the information needed to use ADEMPAS safely and effectively. See full prescribing information for ADEMPAS.ADEMPAS (riociguat) tablets, for oral useInitial U.S. Approval: 2013