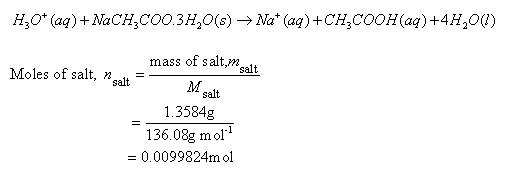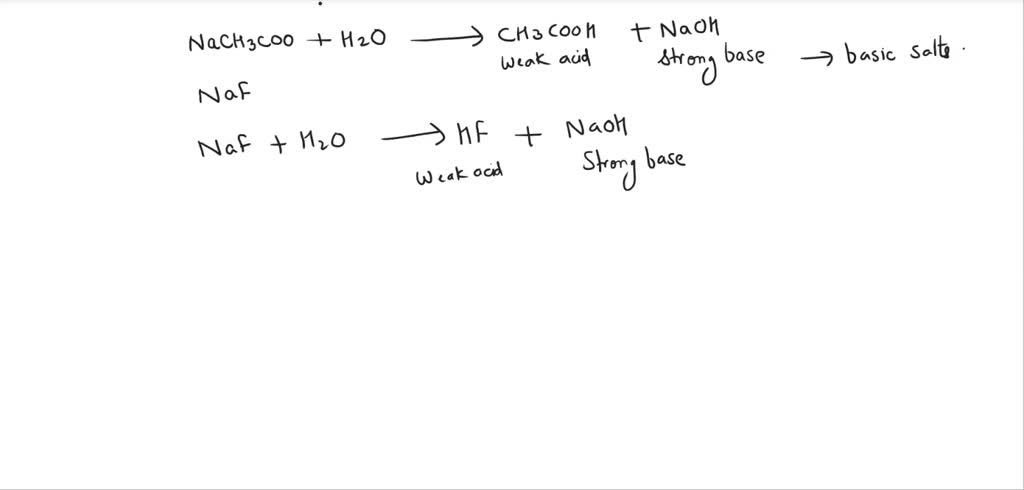
SOLVED: Write chemical equations to show that NaCH3COO and NaF are basic salts. Indicate which of the two salts are more basic in aqueous solutions, and explain why.
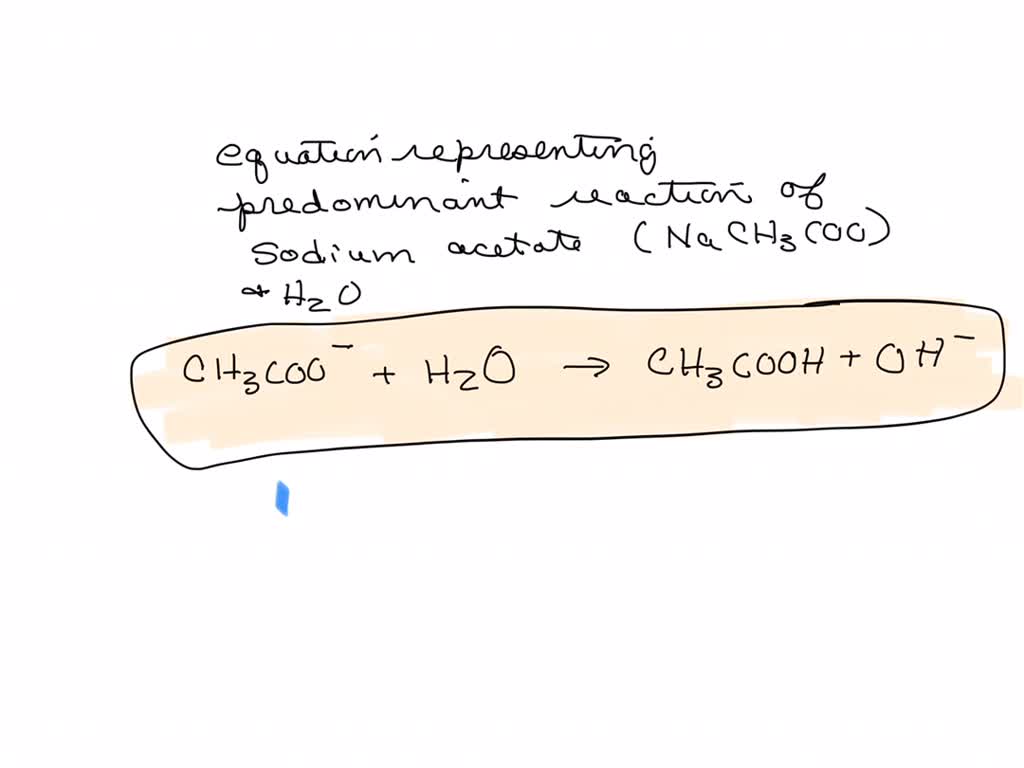
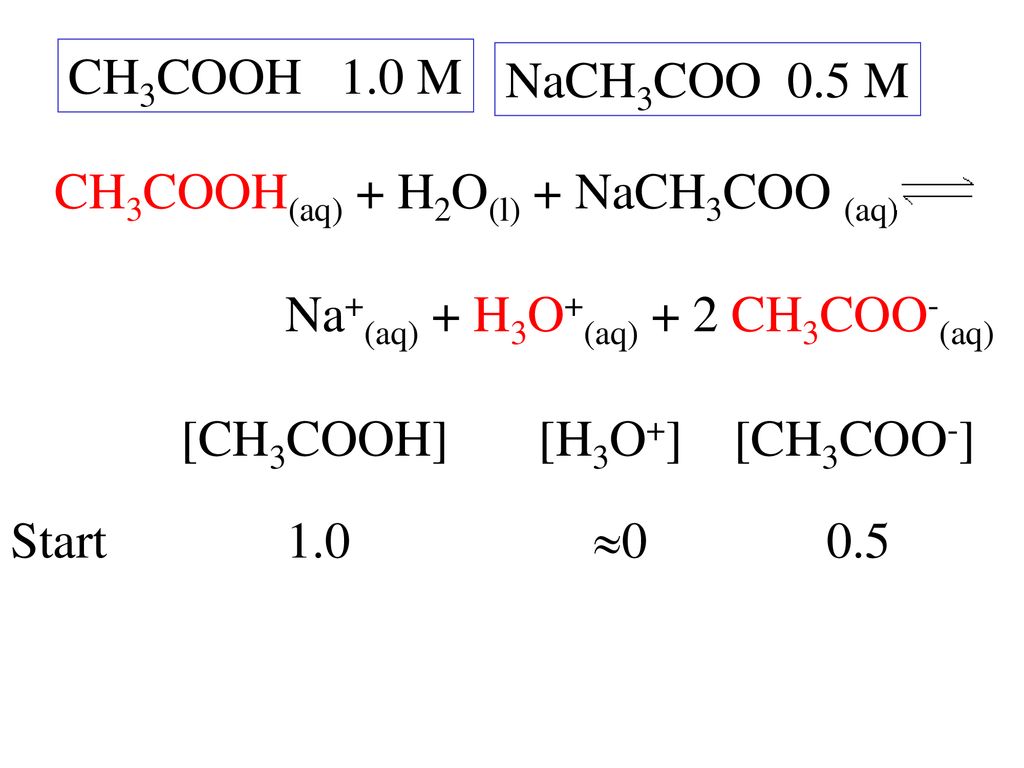
SOLVED: The equation representing the predominant reaction of sodium acetate, NaCH3COO, with water is: CH3COO- + H2O ⇌ CH3COOH + OH- CH3COO- + H2O ⇌ CH3COOH + OH- CH3COOH + H2O ⇌

For sodium acetate solution in water, the given equilibrium reaction occur:CH 3 COO aq + H 2 O l hydrolysis ⇌ CH 3 COOH aq + OH aqWhich of the following describes

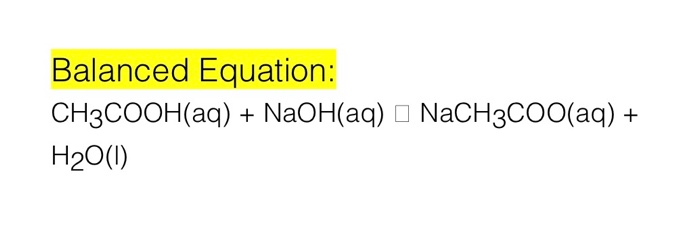
In a reaction 5.3g of sodium carbonate reacted with 6g of ethanoic acid the products were 2.2g of carbon dioxide 0.9g water and 8.2g of sodium ethonoate show that these observations are
Why does the solution of sodium acetate give more concentration of Hydroxide ion? Shouldn't the number of Hydroxide ion and hydrogen ion be equal? - Quora

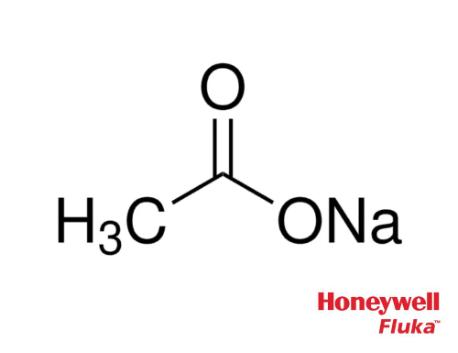
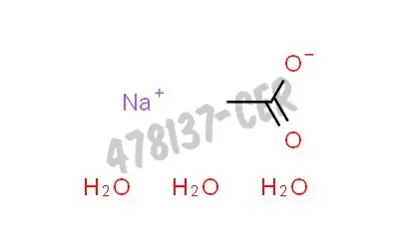
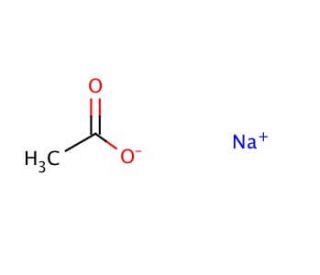
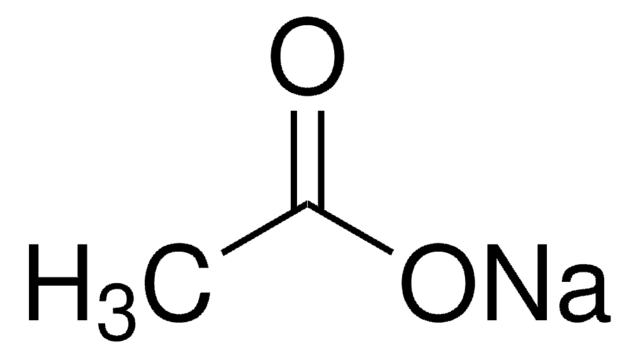
Sodium Acetate(CH3COONa) - Structure, Properties, Preparations, Uses, Important questions, FAQs of sodium acetate.