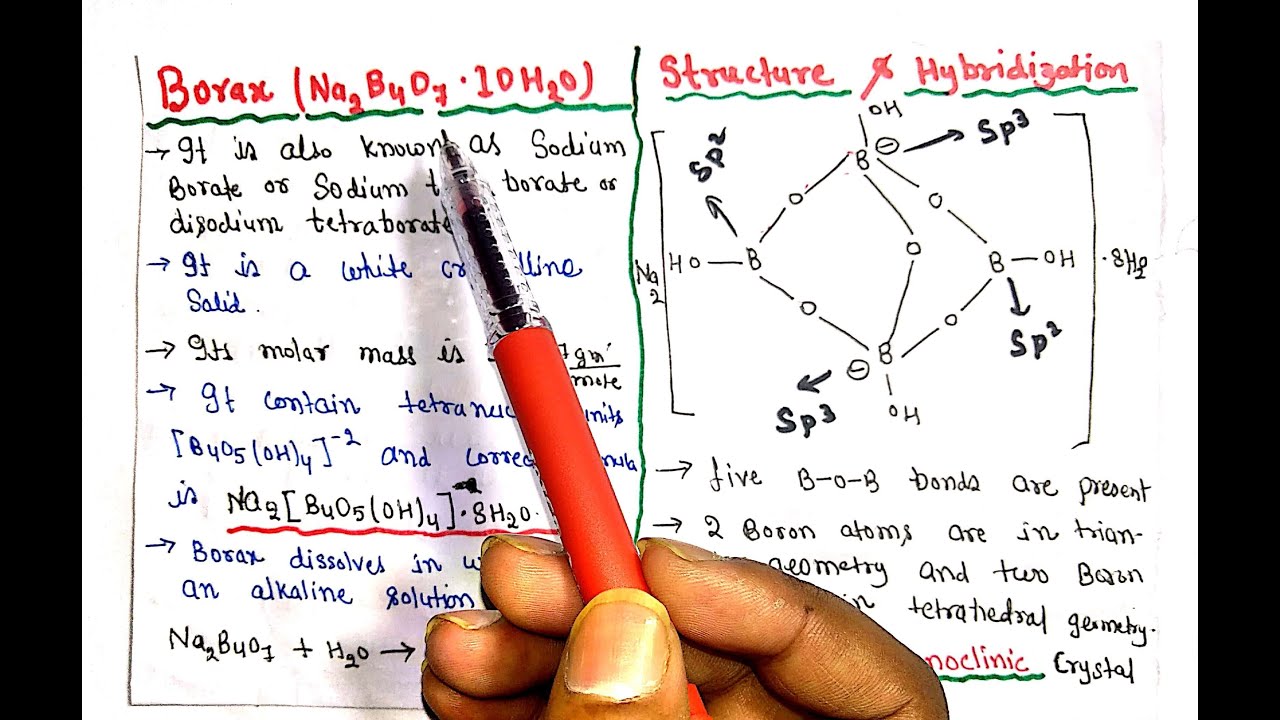
Borax (Na2B4O7•10H2O) || Properties || Structure & Hybridization || Preparation & Heating effect - YouTube

Eutectic Temperature, Density, and Solubility of H3BO3–H2O, Na2B4O7–H2O, and NaBO2–H2O Binary Systems | Journal of Chemical & Engineering Data

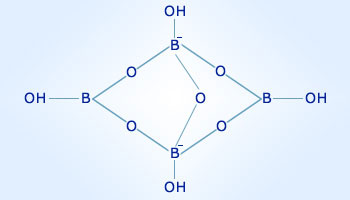
Borax has the formula Na_{2}B_{4}O_{7}. 10H_{2}O. It is a strong base in aqueous solution because OH^{-} ions are produced by reaction with water.(B_{4}O_{7}^{2-}+7H_{2}O rightarrow 4H_{3}BO_{3}+2OH^{-}).How many grams of borax is necessary to
Calculate the % of boron (B) in borax Na2 B4 O7 . 10H2O. (H = 1, B = 11, O = 16, Na = 23) - Sarthaks eConnect | Largest Online Education Community

BORON & ITS COMPOUNDS 2. By passing electric discharge at low pressure through a mixture of BCl 3 or BBr 3 and excess of hydrogen. 3. By reacting. - ppt download




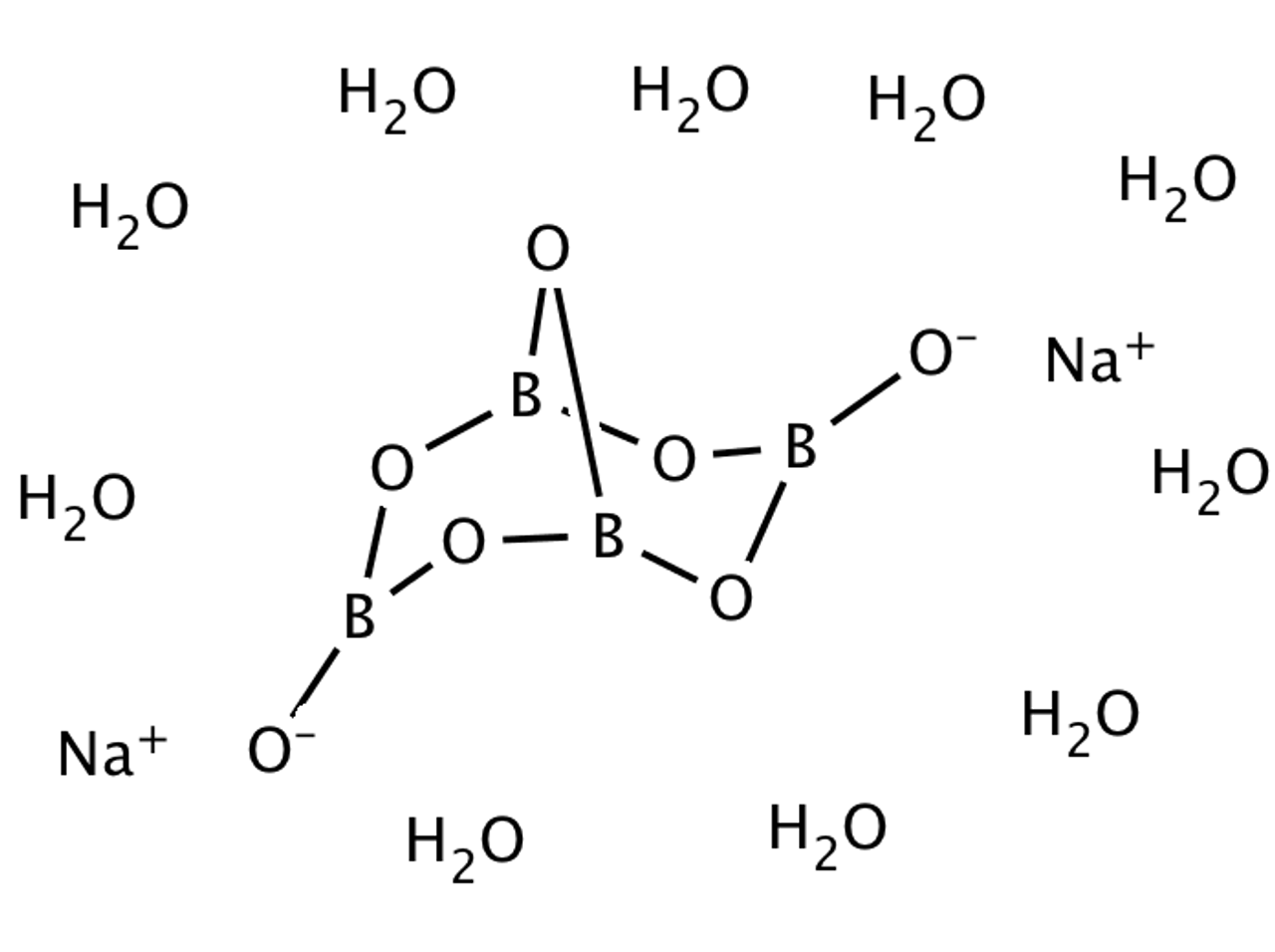

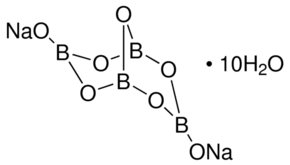







![Alchemy Alphabet: BORAX, also: Tinkal. Sodium... - Stock Illustration [76297336] - PIXTA Alchemy Alphabet: BORAX, also: Tinkal. Sodium... - Stock Illustration [76297336] - PIXTA](https://t.pimg.jp/076/297/336/1/76297336.jpg)