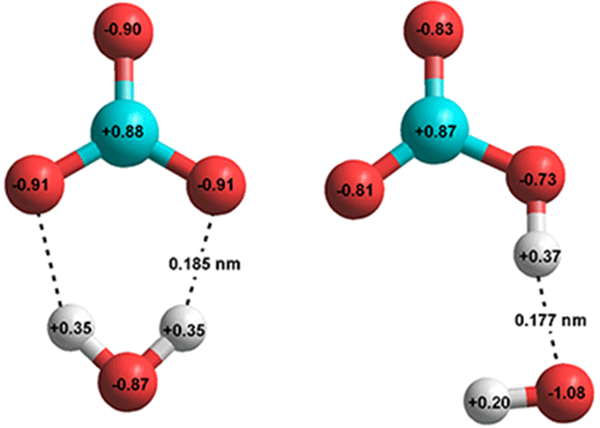
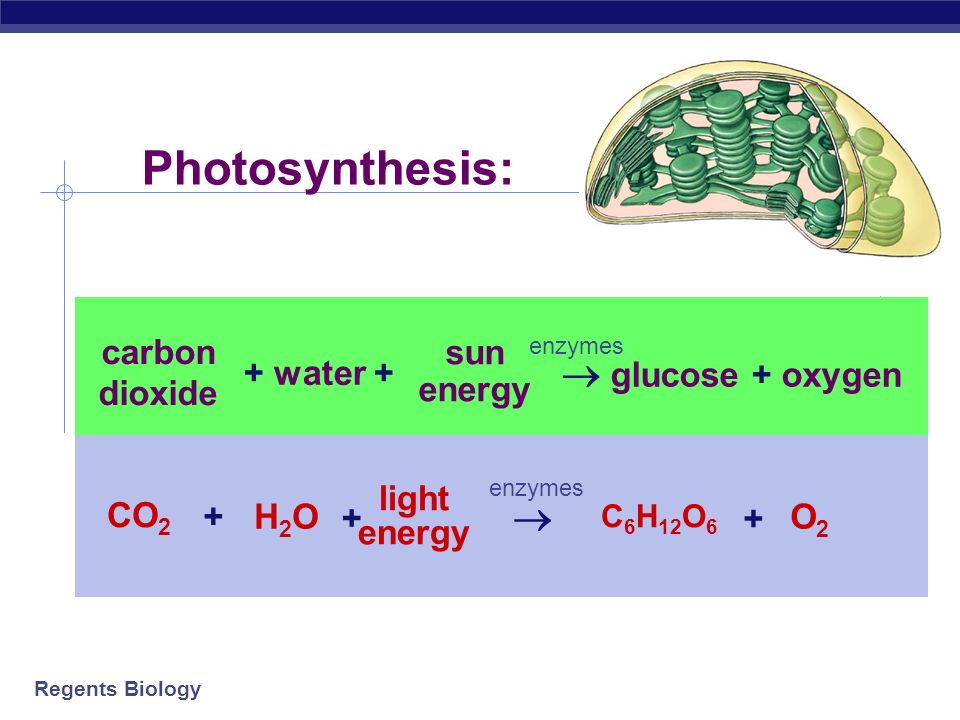
SOLVED: 11. Which correctly shows the inputs and products of photosynthesis? Sunlight Glucose -> H2O + 02 + CO2 Sunlight + CO2 + 02 -> Glucose H2O Sunlight + H2O + C02 -> Glucose + 02 Sunlight + H2O + 02 -> Glucose CO2

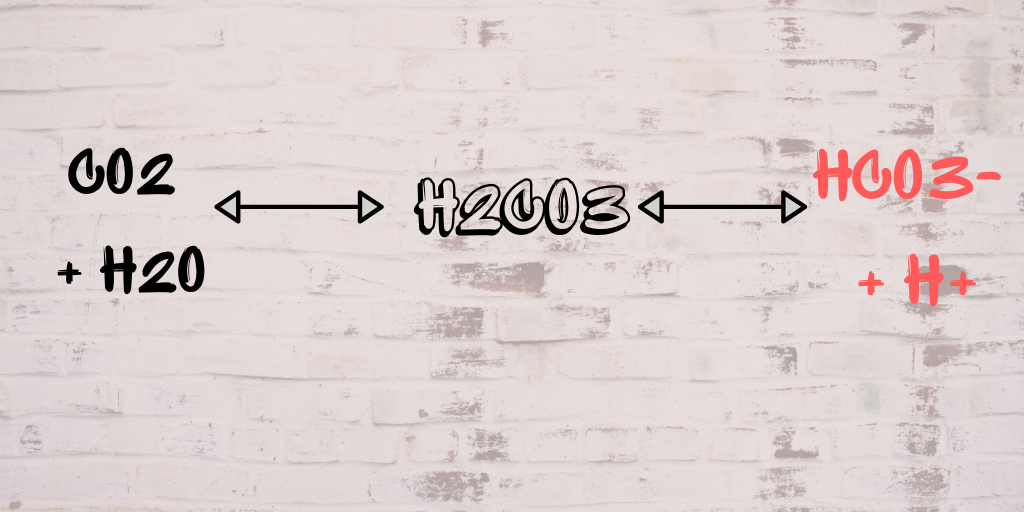
CO2+H2O=H2CO3 balance the chemical equation @mydocumentary838. co2+h2o=h2co3 balance the equation. - YouTube

Reaction of CO, H2O, H2 and CO2 on the clean as well as O, OH and H precovered Fe(100) and Fe(111) surfaces - Catalysis Science & Technology (RSC Publishing)

QM-optimized structures of CO2-water clusters with various numbers of... | Download Scientific Diagram

Water (H2O) and carbon dioxide (CO2) both have one central atom with two atoms bonded to it. However, one is a polar molecule and one is not. Draw the Lewis structure for

Write fully balanced equations for the following:(a) CO2 + H2O(b) Ca(OH)2+ CO2(c) SO2 + H2O(d) P205 + H2O(e) - Brainly.in

Summary of the reactions between carbon dioxide (CO2) with water (H2O)... | Download Scientific Diagram


![Complex mixtures: H2O-NaCl-CO2 [The world of fluid inclusions] Complex mixtures: H2O-NaCl-CO2 [The world of fluid inclusions]](https://fluid-inclusions.univ-lille.fr/res/flinc-11.png)