SOLVED: Write equation for the reaction ammonium acetate (CH3COONH4) with water. What will be the pH of its aqueous solution? Calculate the grams of KHP (potassium hydrogen phthalate) needed to react with

SOLVED: 15.84 When ammonium acetate dissolves in water, both the resulting ions undergo proton transfer reactions with water, but the net reaction can be written without using water: NH4+(aq) + CH3COO-(aq) â†'

Green Synthesis of 1,4‐Dihydropyridine Derivative in Water - Isomura - 2018 - ChemistrySelect - Wiley Online Library

Solubility of H2S in (H2O + CH3COONa) and (H2O + CH3COONH4) from 313 to 393 K and at Pressures up to 10 MPa | Journal of Chemical & Engineering Data

Which of the following has highest value of pH:- 10.1M NaCl 2 0.1M NH4CI 3 0.1M CH3COONa 4 0.1M CH3COONH4
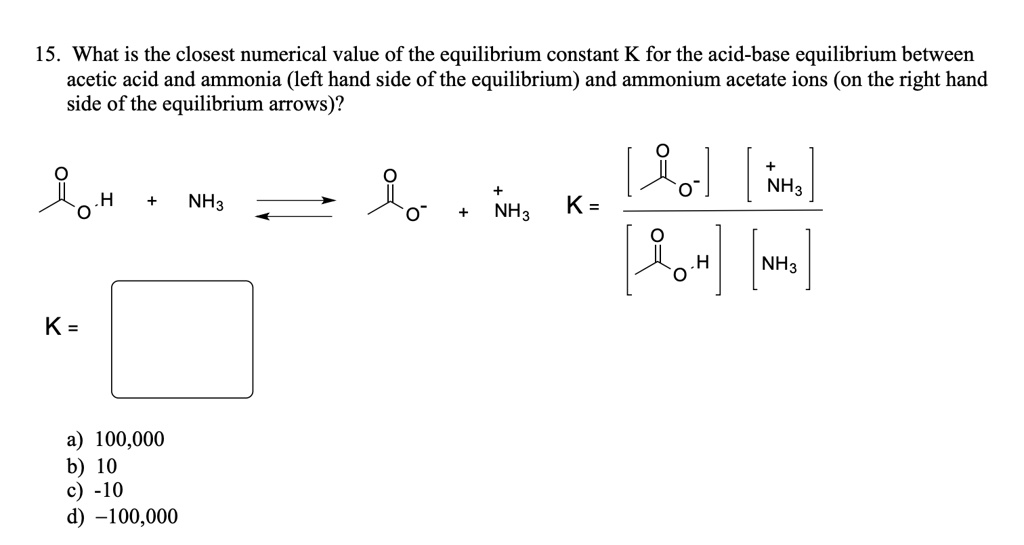
SOLVED: 15. What is the closest numerical value of the equilibrium constant K for the acid-base equilibrium between acetic acid and ammonia (left hand side of the equilibrium) and ammonium acetate ions (

Formation of adenine from CH3COONH4/NH4HCO3—the probable prebiotic route for adenine - ScienceDirect
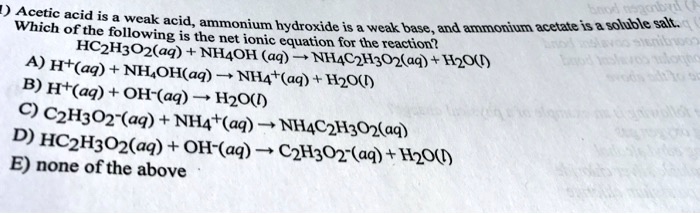



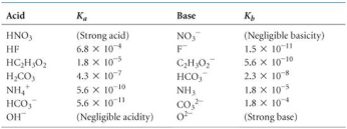

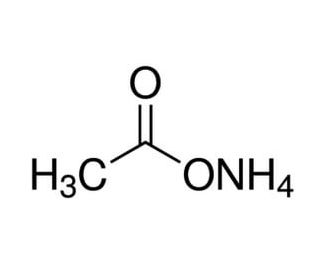


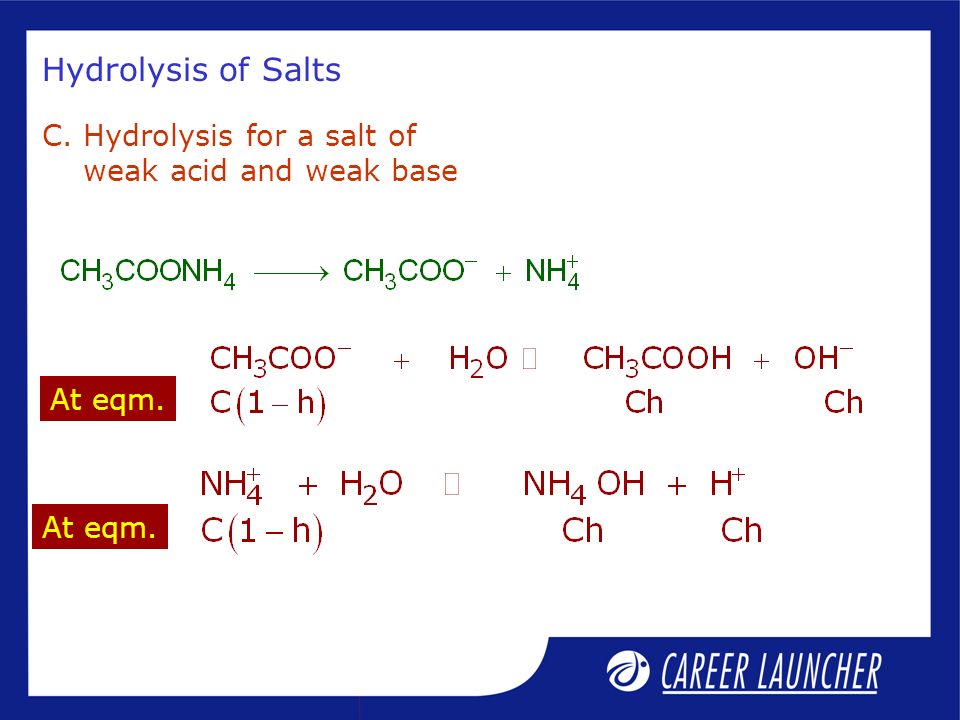
![Welcome to Chem Zipper.com......: [3] CATIONIC AS WELL AS ANIONIC HYDROLYSIS: Welcome to Chem Zipper.com......: [3] CATIONIC AS WELL AS ANIONIC HYDROLYSIS:](https://blogger.googleusercontent.com/img/b/R29vZ2xl/AVvXsEjJbWjsn-LBqxX4y74j0W2DM5MJz-FPpzBl0Sn2S3KZpbegpZvPApc4JmvPT4I3cwFInFG4BZE3E4x2cYaQys3MI3C6K0Py8tIkE3hWaKVpxk-jBlgrGQPHBLUZ4XcVBrz1C1IGUAtPybQ/s1600/SH21.PNG)
![Welcome to Chem Zipper.com......: [3] CATIONIC AS WELL AS ANIONIC HYDROLYSIS: Welcome to Chem Zipper.com......: [3] CATIONIC AS WELL AS ANIONIC HYDROLYSIS:](https://blogger.googleusercontent.com/img/b/R29vZ2xl/AVvXsEgL8SQhnjKci8Sd_t_-hwKS6TbUzdVEN36ZVX7jzyaaImcB0R3-pjiQk5I7lhygJIO2mioVSY0269QkoVgPgOTPfaPDfCu_McwLwOKS5MWlvzzjd4qRRjyHV5vZto0d67HeHHMAvK9k81o/s1600/SH28.PNG)